INDEX
다니엘 러더퍼드Daniel Rutherford
질소는 -195.8 ℃까지 기체를 유지합니다. -210 ℃까지 액체입니다. -210℃를 초과하면 얼음이 고체화합니다. 저렴한 냉각제로 많이 이용되고 있습니다.
액체 질소는 냉각제로 자주 사용되지만, 냉각 가스 소스로도 이용되고 있습니다.
질소의 발견 자의 특정은 어렵습니다.
1772 년 스코틀랜드의 물리학 자 다니엘 러더 포드 (1749-1819)가 질소를 별도로 분리하여 그 안에 생물을 넣으면 질식 해 죽을에서 noxious air (유독 공기)라고 명명했습니다.

칼 빌헬름 셸레Karl Wilhelm Scheele
비슷한시기에 스웨덴의 화학자 카를 빌헬름 세레 (1742 – 1786)와 영국의 화학자, 물리학 자 헨리 카벤디쉬 (1731 – 1810)도 별도로 분리 된 것으로 알려져 있으며, 세레 산소를 ‘불의 공기’, 질소를 “안 공기 ‘라고 명명했습니다.

헨리 카벤디쉬Henry Cavendish

앙투안 라부아지에Antoine-Laurent de Lavoisier,
질소 원소임을 발견 한 것은 프랑스 앙투안 라부아지에 (1743-1793)에서 프랑스어로 “살 수없는 것”이라는 의미의 “azote”라고 명명했습니다.

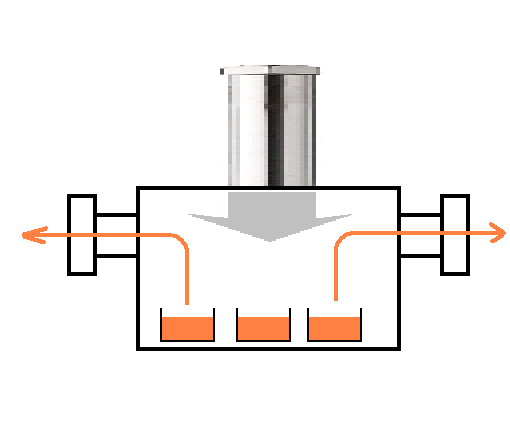
액체 질소를 건설에는,
(1) 공기를 압축하여 액체 공기를 만듭니다.
(2) 그 후 기화시켜 가면, 기화 온도의 차이에 따라 먼저 끓는 조치 183 ℃의 산소가 기화합니다.
(3) 끓는 조치 196 ℃의 질소가 액체로 남아 있습니다.
이와 같이하여 산소를 취한 나머지 것이 액화 질소입니다. 약간의 아르곤이 불순물로서 잔류합니다.
액체 질소는 생체 조직에 부착하면 쉽게 동상을 일으키는 원인이되고 또한 밀폐 공간에서 급격히 기화 시키면 산소 결핍증에 빠지는 때문에 신중한 취급이 필요합니다.
 HEAT-TECH 최선의 기술 온라인(on-line) 샵
HEAT-TECH 최선의 기술 온라인(on-line) 샵