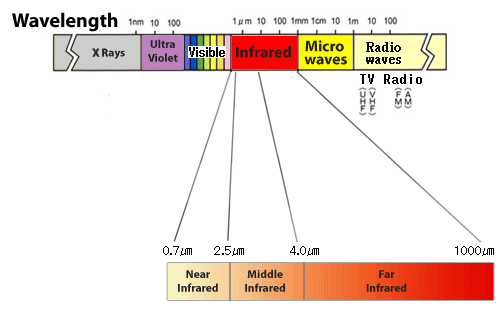
雖然不可見,但由於具有使物體變熱的光而存在於“紅色以外”,因此被稱為“紅外線”。
紅外線是與“ X射線”,“紫外線”,“可見光”,“微波”,“射頻波”等相同的“電磁波”。
電磁輻射(EM輻射或EMR)是輻射能的一種形式,通過光子波粒子在空間中傳播。 在真空中,它以特徵性的速度(通常是直線)傳播,即光速。 EMR被帶電粒子發射和吸收。 作為電磁波,它既具有電場分量又具有磁場分量,它們彼此以固定關係振盪,彼此垂直,並且垂直於能源和波的傳播方向。
EMR的特徵在於其波的頻率或波長。 電磁頻譜按頻率遞增和波長遞減的順序,由無線電波,微波,紅外線,可見光,紫外線,X射線和伽馬射線組成。 各種生物的眼睛感覺到的EMR頻率有些變化,但相對范圍較小,稱為可見光譜或光。 更高的頻率對應於每個光子所承載的更多能源。 例如,單個伽馬射線光子所載能源遠遠大於單個可見光光子。
電磁輻射與電磁場相關,電磁場可以自由地傳播自己,而不受產生它們的移動電荷的持續影響,因為它們已經與這些電荷保持了足夠的距離。 因此,EMR有時稱為遠場。 用這種語言,近場是指在電荷和直接產生電荷的電流附近的電磁場,例如具有簡單磁體和靜電現象的電磁場。 在EMR中,磁場和電場分別由另一種類型的磁場的變化引起,從而以波的形式傳播。 這種緊密的關係確保了EMR中的兩種類型的場都是同相的,並且彼此之間具有固定的強度比,並且每個場的最大值和節點都位於空間的同一位置。
EMR通過不斷遠離源的空間攜帶能源(有時稱為輻射能)(對於EM場的近場部分而言並非如此)。 EMR還帶有動量和角動量。 這些性質都可以賦予與之相互作用的物質。 EMR在創建時是由其他類型的能源產生的,而在被銷毀時會轉換為其他類型的能源。 光子是電磁相互作用的量子,是所有形式的EMR的基本“單位”或組成。 光的量子性質在高頻(因此具有高光子能源)下變得更加明顯。 這樣的光子比低頻光子更像粒子。
在經典物理學中,當帶電粒子被作用在其上的力加速時,就會產生EMR。 電子負責大多數EMR的發射,因為它們的質量很輕,因此很容易通過各種機制加速。 快速移動的電子在遇到力區域時會被最迅速地加速,因此它們負責產生自然界中觀察到的大部分最高頻率的電磁輻射。 量子過程還會產生EMR(例如原子核經歷伽馬衰變時)以及過程(例如中性介子衰變)。
EMR對生物系統(以及在標準條件下對許多其他化學系統的影響)取決於輻射的功率和頻率。 對於較低的EMR頻率直至可見光(即無線電,微波,紅外線),在這種條件下對細胞以及對許多普通材料的損害主要取決於加熱效果,因此取決於輻射功率。 相比之下,對於紫外線及更高頻率的更高頻率的輻射(即X射線和伽馬射線),由於單光子具有 這樣的高頻EMR會化學破壞單個分子。
James Clerk Maxwell首先正式提出了電磁波。 這些後來被海因里希·赫茲證實。 麥克斯韋(Maxwell)導出了電磁方程的波形,從而揭示了電場和磁場的波狀性質及其對稱性。 由於通過波動方程預測的EM波的速度與測得的光速一致,因此Maxwell得出結論,光本身就是EM波。
根據麥克斯韋方程,空間變化的電場總是與隨時間變化的磁場相關。同樣,空間變化的磁場與電場中隨時間的特定變化相關。在電磁波中,電場的變化總是伴隨著磁場中一個方向的波,反之亦然。兩者之間的這種關係是在沒有一個類型字段引起另一個的情況下發生的。而是它們以時間和空間變化同時發生並以相對論相互聯繫的相同方式發生(實際上,磁場可能被視為電場的相對論性畸變,因此此處空間和時間變化之間的密切關係遠大於打個比方)。這些場一起形成傳播的電磁波,該電磁波移入太空,不再需要再影響信號源。通過電荷的加速以這種方式形成的遙遠的電磁場攜帶著隨其“輻射”通過空間的能源,因此稱為“空間”。