INDEX
- What is the Liquid Crystal?
- The history of the Liquid Crystal
- Material for the Liquid crystal display (LCD)
- Physical properties of the LCP (typical)
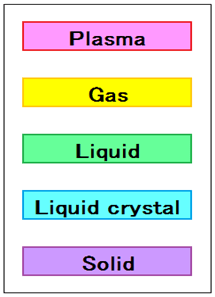
What is the Liquid Crystal?
The liquid crystal is a substance that is an intermediate state or such conditions, liquid and solid (crystalline).
At present, those that express the liquid crystal structure is not found in other than the resin.
The liquid crystal field of located in the development stage, followed by the new discoveries and inventions.
Intermediate state between solid and liquid are the liquid crystal and the flexibility crystal.
Liquid, although there is no regularity in the three-dimensional position is a state where there is regularity in the orientation of the particles.
Plastic crystal, although there is a regularity in the three-dimensional position, is a state where there is no regularity in the orientation of the particles.
As a typical compound of Flexibility crystal, Carbon Tetrachloride, Cyclohexane and Fullerene.
| Regularity | Solid | Liquid crystal | Flexibility crystal | Liquid |
|---|---|---|---|---|
| 3-D position | ○ | × | ○ | × |
| Orientation of the particles | ○ | ○ | × | × |
Liquid crystal polymer
That of the resin expressing the liquid crystal structure is called Liquid Crystal Polymer or Liquid Crystal Plastic(LCP).
Liquid crystals are classified in Thermotropic Liquid Crystal and Lyotropic Liquid Crystal on the basis of the expression format.
Temperature transition type (Thermotropic) LCP
Thermotropic liquid crystal changes to “Liquid Crystal” between the melting point and the clearing point.
Arranged linear molecules regularly in the molten state to a liquid crystal-like properties, it is defined as a thermoplastic resin.
Liquid crystal resin that is currently used industrially is mostly this type.
Concentration transition type (Lyotropic) liquid crystal
Lyotropic liquid crystal, the front to become a complete solution when dissolved into something of a liquid, will change to “Liquid Crystal” in a certain concentration range.
It becomes a liquid crystal stimulation of dissolution, it can be seen when dissolved a certain surfactants in water at high concentrations.
By hydrophilic and hydrophobic moieties gather each, you can massive structure over the entire solution.
Liquid crystal has many aspects in the base of 4 systems.
(1). 0-dimensional center of gravity order system
Nematic phase
Chiral nematic phase
Discotic nematic phase
Cholesteric phase
(2). Primary periodic structure system
Smectic phase (the abbreviation Sm phase)
There are manyphase , such as SmA SmB and SmC, moreover newer discovery continues.
The banana type smectic phase (the abbreviation B phase)
There are many phase, such as B1, B2 and B3, moreover newer discovery continues.
(3). 2-dimensional periodic structure system
Discotic columnar columnar phase
(4). 3-dimensional periodic structure system
Cubic phase
Blue phase
*Variety of conditions is present in the liquid crystal, new discoveries have followed.
The history of the Liquid Crystal
The discovery of the lyotropic liquid crystal

Rudolf Ludwig Karl Virchow (Oct 13, 1821- September 5, 1902) Pathologist of German, discoverer of Leukemia
1854, R. Firuhyo is brought into contact with myelin and water, which is a living body of the nerve tissue, we found a lyotropic liquid crystal.
The discovery of thermotropic liquid crystal

Friedrich Richard Reinitzer (Feb 25, 1857 – Feb 16, 1927) Austrian botanist, chemist
1888, F · Reinitzer is, by chance during the study of plant physiology Institute of Prague, ester compound cholesteryl benzoate of cholesterol and benzoic acid was noticed that melting twice.
When the usual solid to heat the crystal, it becomes liquid at a certain temperature (melting point).
However, when heating the crystals of cholesteryl benzoate becomes a cloudy liquid at 145.5 ℃, it is even more became a clear liquid at 178.5 ℃ on heating.
In other words, he has discovered that there are two of the melting point in cholesteryl benzoate.
F · Reinitzer thought this wonder was asked to further investigation to O · Lehmann.

Otto Lehmann (Jan 13, 1855 – June 17, 1922) German physicist
Lehmann has developed a heated polarized light microscope is a time of state-of-the-art equipment.
Lehmann was observed this mysterious liquid cholesteryl benzoate in heated polarized light microscope, it has confirmed certain phenomenon.
Originally in cholesteryl benzoate of the liquid state we found that there is the nature of the birefringence that refracts light with a solid crystal in two directions.
Birefringence those with anisotropy, that is because the phenomenon does not occur only in crystals has been known since then.
Even though the state of the liquid is thought to have properties such as crystal, he has written a report called “About flowing crystal”.
In the wake of this, various materials have been analyzed.
1911 France of Charles Victor Morgan found a liquid crystal alignment by rubbing.
1922 French Georges Friedel has established a three-classification of liquid crystal called “smectic nematic cholesteric”, and that the liquid crystal and intermediate = liquid crystal, it was named “liquid crystal”.
1963 US RCA Corp.’s R · Williams I’ve discovered that the change in opacity is transparent liquid crystal when a voltage is applied to the liquid crystal.
1968 US RCA Corp. found a phenomenon in which changes the voltage of the DC and low-frequency crystal is milky white was a transparent when applied to a nematic liquid crystal (liquid crystal filamentous pattern), “dynamic scattering effect” and named after to the liquid crystal application sit ushered in the road.
1973 Japan of SHARP is put to practical use the dynamic scattering type liquid crystal (DSM) as a display body, have been developed calculator with the world’s first liquid crystal display device, (LCD Display) are utilized in a variety of equipment.
1974 US Eastman Kodak developed and on the market to “Xydar”, was aimed at improving the heat resistance of polyethylene terephthalate.
1979 Japan’s Sumitomo Chemical (now Sumitomo Chemical) has developed the “Ekonoru (now SUMIKASUPER)”, has been improved heat resistance so as to correspond to the surface mount technology (SMT).
1984 US Celanese has developed the “Vectra”, it was expanding the coverage of the mechanical parts.
Material for the Liquid crystal display (LCD)
The optical shutter by the liquid crystal
Liquid crystal, simple molecular motion by such as voltage and magnetic force, the way through the light will change.
When use this property, it will be able to make the shutter of the electrically controllable light.
That is the Twisted Nematic Liquid Crystal.
The combination of liquid crystal and a polarizing filter with a twisting nature, but usually is passed through the light and will not transmit light lost is twisted when a voltage is applied, and is intended to act as a light shutter.
The display developed in this manner is referred to as TN type, STN LCD of improved type was much used in one time of the laptop.
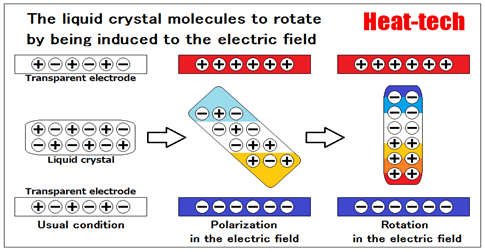
The liquid crystal in replies to an electric field
The word nematic (nematic) comes from “nemato-” means “filamentous” “nematode”.
Often seen turtle shell is continuous backbone to the liquid crystal molecules of filamentous, it has a property that the electrons move relatively freely contained therein, are especially made to move more easily clause along the length of the molecule.
The addition of an electric field from outside Therefore, bias the electrons closer to the positive pole, plus the length of the molecule, a negative polarization occurs.
Then, in the minus plus side of the electric field of the molecule, the negative side of the molecule is attracted to the positive of the electric field, the molecules will direction to rotate the electric field.
This is the orientation by the electric field of the liquid crystal.
LCD combines the crystallinity of the liquidity and solid liquid.
Molecules of the liquid crystal can be moved freely like molecules of the liquid, but for certain direction has property of regularly arranged like the atoms in the solid crystals.
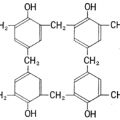
Liquid crystals are used in LCD has an elongated shape. For example, the molecules of the nematic liquid crystal, which is called the 5CB has a structure in which the hard part and the carbon to which benzene rings are connected as shown in the following figure is made from soft part that led to the linear.
Positive-type liquid crystal
Dielectric constant: small in a direction perpendicular to the large major axis in the longitudinal direction
Positive-type liquid crystal is used in TN type, IPS type.
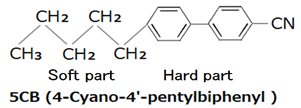
The liquid crystal molecules has a fluidity such as liquid since there is the soft part can be arranged regularly because there is a hard part.
Applying a voltage to the liquid crystal, by a cyano group (-CN), and polarization made has a portion biased portion and negative charges biased positively charged in the liquid crystal molecules.
Therefore, the liquid crystal molecules are regularly arranged in the direction of the electric field. It is not arranged regularly when not applying a voltage.
Negative-type liquid crystal molecules
Dielectric constant: The smaller the longitudinal direction, is greater in a direction perpendicular to the longitudinal axis
Negative type liquid crystal is used in a VA-type.
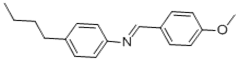
(4-Methoxybenzylidene)-(4-Butylaniline)(MBBA)
| Material | Melting point (℃) | Clearing point (℃) |
|---|---|---|
| MBBA | 22 | 47 |
| 5CB | 23 | 35 |
| 8CB | 22 | 34 |
The nature of the structure
Nematic liquid crystal will move freely without the limitation position of the molecule, to move molecules against the force from the outside and then you easily deformed, it does not return to the original on their own Once deformation.
Always flowing in the same way as ordinary liquid, it is the extent that slightly against the deformation to have a viscous.
When you try to change the orientation of the molecule in part, and then resistance working force that tries to return to the original.
This is it is not a phenomenon seen in ordinary liquid, yet the resistance force, vertical bending, lateral bending, twisting, etc., it is different depending on the direction of the deformation.
By utilizing such characteristics in injection molding, it is possible to make a tough super engineering plastic.
By dense crystal structure that occurs after molding, there is a rigidity in excess of engineering plastics that are reinforcing fillers even in non-reinforced state.
For low viscosity no entanglement of the polymer cross-liquid crystal property shown at the time of melting and excellent fluidity during molding.
In addition, since the molding shrinkage and linear expansion coefficient is low, it can respond to the thin-walled structure and fine molding.
Such liquid crystal named Liquid Crystal Polymer or Liquid Crystal Plastic (LCP) .
Strictly speaking, the para-hydroxybenzoic acid as a basic structure, because the melting point of the homopolymer would exceed the 600 ℃ and thermal decomposition temperature, the aromatic polyester resin obtained by an ester bond to a variety of components and the linear, crystalline polyester and it has also been said.
Generally, it is produced by a melt polymerization method.
The hydroxy group of the aromatic hydroxy acid was acetylated by acetic anhydride, etc., and heated to undergo de-acetic acid polycondensation reaction and create a linear structure.
To produce a relatively low molecular weight polymer in the melt polymerization method, there is also a method to further polymerization in this solid-phase polymerization method.
LCP improved study of the constituent molecules will proceed to seek a balance between fluidity and heat resistance, there are already more than 20 of the LCP, it has been further developed.
(Type Ⅰ) Polycondensates of the 4,4-dihydroxy biphenol and terephthalic acid and a para-hydroxy benzoic acid
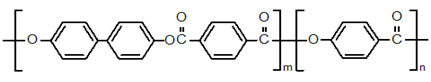
Econol- Sumitomo Chemical
Xydar-Amoco
(Type Ⅱ) Polycondensate of 2,6-hydroxynaphthoic acid and para hydroxy benzoic acid
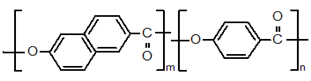
Vectra- Celanese
(Type Ⅲ) Polycondensate of ethylene terephthalate and a para-hydroxy benzoic acid
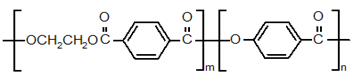
Rodorun – Unitika
Novaccurate-Mitsubishi Chemicals.
Physical properties of the LCP (typical)
| Unit | TypeⅠ | TypeⅡ | TypeⅢ | |
|---|---|---|---|---|
| Specific gravity | – | 1.60-1.70 | 1.62 | 1.62 |
| Tensile strength | MPa | 108 | 206 | 118 |
| Elongation at break | % | 1.3 | 3 | 5 |
| The tensile modulus of elasticity | MPa | 14014 | 9806 | – |
| Flexural strength (23 ℃) | MPa | 137 | 152 | 137 |
| The flexural modulus (23 ℃) | MPa | 12152 | 8820 | 9310 |
| Impact strength (Izod / 6.4t notch) | J/m | 163 | 431 | 392 |
| Hardness (Rockwell) | – | – | R66 | R70 |
| Deflection temperature under load (1.81MPa) | ℃ | 266-320 | 230-240 | 180-210 |
| Linear expansion coefficient (MD direction) | x10-5/℃ | 0.0093 | – | 0.1 |
| Permittivity | 103/106Hz | 3.56/3.10 | 3.6/3.4 | – |
| Dielectric loss tangent | 103/106Hz | 0.0068/0.041 | 0.024/0.018 | – |
| Volume resistivity | Ω・cm | – | 6×1016 | – |
| Arc resistance | sec | 186 | 137 | – |
| Combustion quality | mm | V-0, 1.6 | V-0, 0.8 | – |
| Oxygen index | % | 42 | 35 | – |
| Molding temperature | ℃ | 350-400 | 300-330 | 275-290 |
 HEAT-TECH Best Technology Online Shop
HEAT-TECH Best Technology Online Shop