물질의 온도에 나타난 관찰 할 수있는 열 = 현열 물질의 상태를 변화 시키지만 관찰 할 수없는 열 = 잠열.
현열(顕熱) + 잠열(潜熱) = 전열(全熱)
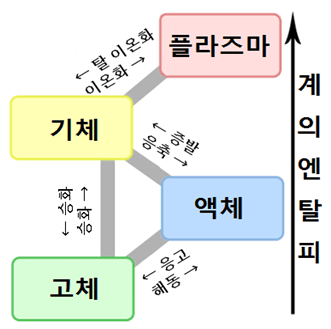
물질의 3 태 상전이
| 전이전의 상 | 전이후의 상 | 현상의 호칭 | 전이점의 호칭 | 전이열의 호칭 |
|---|---|---|---|---|
| 기상 (기체) | 액상 (액체) | 응축 (액화) | (특별히 없음) | 응축열 |
| 고상 (고체) | 승화 (응고, 응결) | (특별히 없음) | (특별히 없음) | |
| 액상 (액체) | 고상 (고체) | 응고 (고화) | 어는점 | 응고 열 |
| 기상 (기체) | 증발 (기화) | 비점 | 증발열 (기화열) | |
| 고상 (고체) | 액상 (액체) | 융해 | 융점 | 융해열 |
| 기상 (기체) | 승화 (기화) | 승화 점 | 승화 열 |
* 증발열은 물질에 흡수되는 열을 나타내고 있고 긍정적 인 응축열은 물질이 방출하는 열이기 때문에 음수 값을 취합니다.
2-9-1.잠열(潜熱)
잠열이란 물질이 고체에서 액체, 또는 액체에서 기체로 상전이 할 때 필요한 열에너지의 총량입니다.
잠열은 융해에 따른 융해열과 증발에 따른 증발열 (기화열)이 있습니다.
잠열의 개념은 조셉 블랙이 도입되었습니다.
블랙은 1761 년에 얼음이 녹는 동안 온도를 변경하지 않고 열을 흡수하는 것을 보여주고 열소 (카로릿쿠)가 얼음 립자와 결합 것이라고 생각했습니다.
또한 같은 체적의 물과 수은의 온도 상승에 차이가 있다는 사실 등에서 열용량과 비열의 개념을 만들어내는 등, 열 학의 발전에 기여했습니다.

조셉 블랙 (Joseph Black, 1728 년 4 월 16 일 – 1799 년 11 월 10 일) 스코틀랜드인
2-9-1-1.융해열(融解熱)
융해열은 일정한 양의 물질이 고체에서 액체로 상전이 할 때 필요한 열량입니다.
단위는 J / g 또는 J / mol에서 얼음의 융해열은 333.5J / g입니다.
2-9-1-2.증발열蒸発熱 (기화열気化熱)
증발은 액체 상태의 원자 또는 분자가 충분한 에너지를 얻고 기체 상태로되는 과정입니다.
액체의 증발 비등점 이하의 온도에서 일어나고 증기압이 포화 증기압 될때까지 계속 거기서 액상 평형에 도달 종료합니다.
액체의 온도가 끓는점에 도달하면 액체 내부에서도 기화 (종기)이 일어납니다.
여기에서 증발시 액체는 주위로부터 증발열 (기화열)을 필요로합니다.
액체의 표면 장력 (분자간 력)을 극복 열 운동 에너지를 가진 분자가 증발 할 수 있습니다.
즉, 증발하는 분자는 액체 표면에 부착 대한 일 함수를 초과 역학적 에너지를 가지고 있습니다.
따라서 증발은 액체의 온도가 높을수록 표면 장력이 낮을수록보다 빠르게 진행합니다.
증발에 참여하고 그 계의 엔트로피는 증가하고 있으며, 상 변화에 따라 에너지의 유입이 필요합니다.
증발을 계속 시키거나 가속하려면 히터에 의한 열 에너지의 지속적인 공급이 필요합니다.
예를 들어 헬륨의 증발열이 0.0845 kJ / mol과 매우 낮은 것은 헬륨 원자 사이에 작용하는 반 데르 발스 힘이 매우 작기 때문에입니다.
증발열 측정 비등점에서 이루어지고 있지만, 일반적으로 298K (25 ℃)에서의 값으로 보정 된 값이 사용됩니다 (보정에 의한 변화는 측정 오차 이하이므로 무시할 수 있습니다).
단위는 kcal / mol (칼로리 당 두더지)가 사용되고 왔지만 최근 kJ / mol (킬로 줄마다 몰)과 표기가 주류입니다.
또한 증발열을 분자간 력의 측정에 이용하는 경우는주의가 필요합니다.
분자간 력은 기상의 물질로도 작용하기 때문에 실제보다 작은 값이 측정 될 수 있습니다.
특히 금속 기체는 공유 결합 상태로 존재하고 있기 때문에 분자간 힘의 측정은 원자화 열을 측정해야합니다.
2-9-2.현열(顕熱)
현열이란 물질의 상태 (상)을 바꾸지 않고 온도 만 변화시키기 위해 소비되는 열량입니다.
예를 들어 20 ℃의 물을 주전자에 넣고 불에 겁니다.
불에서 열이 야간에 전해져, 20 ℃에서 60 ℃까지 온도가 상승했습니다.
이 때 물이 얻은 열을 현열입니다.
학술적인 정의
“다수의 분자 등으로 구성되어있는 계에서 내부 에너지의 증가와 함께 계의 온도도 함께 상승 할 때,이 미세한 역학 에너지를 현열한다”
 HEAT-TECH 최선의 기술 온라인(on-line) 샵
HEAT-TECH 최선의 기술 온라인(on-line) 샵