INDEX
Brazing with Heater
- About Brazing
- Brazing with Air Blow Heater
- Brazing with Halogen Point Heater
- Brazing with Halogen Line Heater
About Brazing
Brazing is a welding method like soldering that joins metals together.
In brazing, joining is possible without melting the base material.
In this method, the base material is “wet” with the brazing filler material melted by the heat conduction of the base material, and then diffused and joined.
Difference between Brazing and Soldering
Difference in Melting Point of Filler Metal
The distinction between brazing and soldering is determined by the melting point of the filler metal.
Brazing uses a filler material (braze) with a melting point of 450℃ or higher, and soldering uses a filler material (solder) with a melting point of 450℃ or lower.
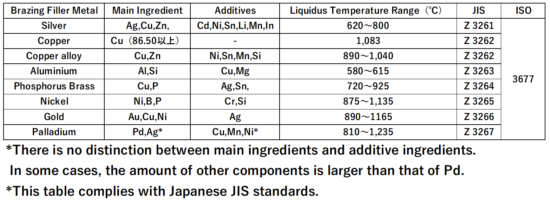
For example, the melting points of commercially available brazing filler metals are higher than 580℃ for aluminum brazing, 735℃ for copper brazing, and 745℃ and 450℃ for silver brazing.
In contrast, the melting point of general lead-containing solder is 183℃, and the melting point of lead-free solder is 217℃.
Difference in Intensity
Brazing is stronger than soldering.
However, the strength of joints is not determined only by the strength of brazing.
In general, the stronger the base metal and the thinner the brazing joint, the stronger the joint.
Features of Brazing
◎Since there is no need to heat the base material to the melting point, there is little thermal effect on the base material, making it easy to join thin and small objects.
◎ It is hard to deteriorate the base material.
◎Possible to join dissimilar materials with different melting points.
[Caution] Brazing with different thermal expansion coefficients causes the brazing layer to tear due to the difference in shrinkage during cooling, so it is necessary to consider the joint method.
◎Because brazing has a lower melting point than the base material, it is possible to remove or rejoin brazed points by reheating.
*When brazing two parts close to each other in two steps, it is possible to avoid remelting the first time by using brazing filler material with different melting points.
◎ Unlike mechanical joining methods such as bolting, airtight and watertight sealing is possible.
◎Complicated shapes with many joints can be joined. (Multi-point batch joining)
◎It is electrically conductive unlike bonding with adhesive.
◎ It does not require as much skill as shielded arc welding, and the work procedure can be learned in a relatively short period of time.
◎By considering the shape of the joint, it is possible to create a joint with strength comparable to that of the base material.
◎ The work can be relatively automated.
About Brazing Filler Material
Brazing filler metal is an additive metal that has a lower melting point than the base material and is used for joining.
It is used by heating the brazing filler metal above its melting point using the heat conduction of the base material, melting it, and diffusing it into the joint by capillary action.
Brazing filler material selection
1.The melting point of brazing is lower than that of the base material
The melting point must be appropriate, and the composition must be such that the parts do not melt together during brazing.
2. Good wettability and moderate fluidity
3. The brazed joint must have the required properties such as mechanical properties such as strength, electrical properties such as electrical conductivity, and corrosion resistance in the operating environment.
4.It should be a material that can be easily machined into wire and plate.
Alloys can also be used as brazing filler metals if they conform to these requirements.
In reality, other factors such as workability, work load, and economic efficiency are also important options, and the types of brazing filler materials that can be used are limited.
Melting point of Brazing Filler Material
If the melting point of the brazing filler metal is low, the heating time can be shortened, which is economical and can prevent deterioration of the base material.
Therefore, rather than using pure metals, there are many brazing filler materials that contain alloy elements to lower the melting point.
For example, the melting point of 100% silver is 961.8℃, but if 28% copper is added and the proportion of silver is 72%, the melting point changes to 780℃. This composition is called a eutectic composition.
The melting point can also be lowered by adding a low-melting element. Brass brazing is made by adding low melting point zinc to copper.
Zinc is added in addition to copper in most silver brazes. There are also silver brazing filler metals with tin or nickel added to this base.
However, if the proportion of low-melting additive metals is too high, the braze may become brittle and poorly workable.
Principle of Brazing
Oxygen in the air reacts with the base material atoms to form an oxide film on the surface of many metals.
Even if molten brazing filler metal is brought into contact with a metal surface on which an oxide film has been formed, the brazing metal atoms and the base metal atoms cannot attract each other.
A force called intermolecular force acts between molecules to attract each other.
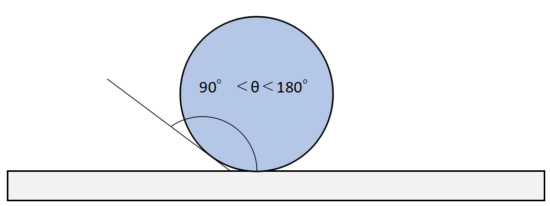
If there is an oxide film, there is no attraction between the brazing filler material atoms and the base material atoms. This state is said that the brazing filler material does not wet the base material.
It is like a new umbrellanew umbrella with water droplets repelled on it. The brazing filler material flows because there is no intermolecular force.
In order to perform brazing, it is necessary to wet the base material with the melted brazing filler material, and for that reason, it is necessary to remove the oxide film.
There are two ways to remove this oxide film: one is to use a reducing atmosphere such as hydrogen to remove the oxygen in the oxide, leaving only metal atoms, and the other is to use flux.
When the melted flux comes into contact with the oxide film on the surface of the base material, oxygen is removed from the oxide film, leaving only the metal atoms of the base material.
The action of this flux chemically removes the oxide film,
The metal surfaces of the brazing filler metal and the base metal can be in direct contact.
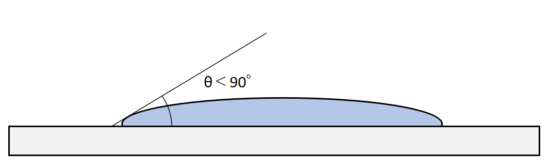
When the melted and liquefied brazing filler metal flows in that state, the metal atoms of the base material and the metal atoms of the brazing filler metal exert intermolecular force and bond.
This state is called the brazing filler metal wetted to the base metal.
It is like a worn out umbrella, which rain permeates and diffuses an umbrella that has lost its water-repellent effect.
Even after the brazing filler metal is wet with the base metal, if the heating is continued, t
The brazing filler metal atoms will penetrate between the atoms of the base metal, creating areas where the brazing atoms and the base metal atoms are mixed.
This layer is called an alloy layer (diffusion layer).This alloy layer strengthens the joint.
About Flux
The action of flux is to remove the oxide film on the metal surface, and the flux chemically reacts with the oxide to create a product (metal salt) that dissolves and removes.
When steel or copper is brazed with borax or boric acid as a flux, the oxide film is dissolved and removed by the following reactions.
FeO (Iron Oxide) + Na2B4O7 (Borax) ⇒ Fe(BO2)2+2NaBO2
CuO (Copper Oxide) + 2H3BO3 (Boric Acid) ⇒ Cu(BO2)2+3H2O
Flux does not have the effect of removing thick oxides such as rust on the surface of the base material, coating materials, oils and fats, dirt, etc.
Therefore, it is necessary to remove these foreign substances before brazing.
If foreign matter such as oil or scale adheres to the base material, the flux will not work properly.
These foreign substances can be sufficiently removed by degreasing and polishing.
Also, the melting point of flux is generally 50℃ lower than that of brazing filler material.
Brazing Heating
Heating work is indispensable for brazing.
Therefore, the thermal effects of heating on the base material cannot be avoided, and structural changes such as oxidation, softening, hardening, and coarsening of the surface of the base material occur.
There are various heating methods for brazing, but the most important factor in all heating methods is temperature control.
The base material near the joint is uniformly heated to the prescribed brazing temperature, and once the braze begins to flow, it is maintained at that temperature until it has completely penetrated the joint.
It is necessary to control the temperature stably by suppressing the temperature change.
Also, since the heating temperature and heating time vary depending on the shape of the base material and brazing filler material, it is necessary to determine the optimal heating conditions.
the Points of Brazing Heat
◎The brazing part is not directly heated, but the base material near the joint is heated, and the conductive heat melts the braze.
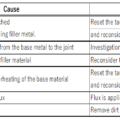
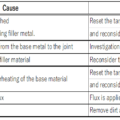
◎When the base material is overheated, a thick oxide film is formed on the surface, making it difficult for the flux to remove the oxide film, and the braze will not wet the base material.
Furthermore, overheating increases the thermal effect on the base material, or it reaches the melting temperature of the base material and melts.
◎If the heat capacity and wall thickness are different, heat the larger and thicker first.
◎ Judge the brazing temperature based on the color of the base material and the degree of flux melt. Brazing addition heat with a halogen heater uses a radiation thermometer in addition to these, and the heating temperature can be managed by feedback control.
◎Be careful not to use more brazing filler metal than necessary, as the brazing filler metal will enter the gaps between joints due to capillary action.
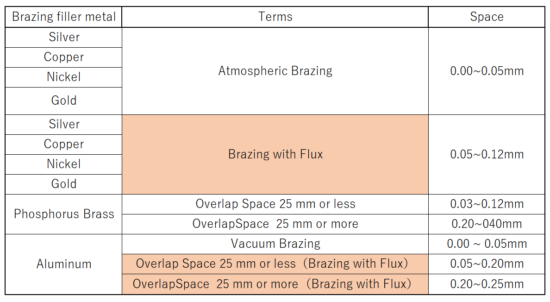
About Joint Spacing
In brazing, the brazing filler material flows into the gaps due to capillary action. If the gap is too wide, the brazing filler metal will flow into the side with the smaller gap, or the gap will remain even after brazing, resulting in poor joints.
It is necessary to figure out the proper spacing and braze.
Types of Brazing and Appropriate Joint Spacing
 HEAT-TECH Best Technology Online Shop
HEAT-TECH Best Technology Online Shop